This section is research and experimentally motivated. It contains some far-fetched hypotheses and captures related information. Feel free to brainstorm along, keeping in mind these barely meet the level of hypothesis. Do not confuse this section with generally well validated Really Correct material. Experimental and Developmental. If you have a suggestion for further research, please Contact and Comment.
President Diagnosed With COVID-19
It was reported at about 5PM EST October 1 that President Trump was experiencing “mild” COVID-19 symptoms and had tested positive. It could be guessed that symptoms were present earlier that day so a guestimate of symptom appearance would be 12PM EST October 1. “Trump is receiving a two-antibody combo drug that’s currently in late-stage studies from Regeneron Pharmaceuticals Inc.” per a news article October 2. Backing up 4 days, the early exposure time would be Monday 12PM EST September 28. Given this timetable, he probably already has a natural IgM ramp that is nearing peak and an IgG ramp that is beginning. In most cases, peak symptoms will occur about 3 to 4 days after initial symptoms, so his worst day may be Sunday October 4th or Monday October 5th. By Wednesday October 7th, he will likely be informing the world he is feeling better. If the antibody drug has efficacy, then this timetable would likely be accelerated. Because he is stated to already be experiencing mild fever as of October 2, the date of initial symptoms might actually be a day earlier than estimated based on press-coverage (earliest symptoms of fatigue and headache are often easily dismissed). If so, that would move the timeline 1 day earlier. If he does not exercise due to hospitalization, this could move the timeline 2 days later (or even much longer). It is likely he will stubbornly do what he wants to do, which would intelligently include light exercise. Reports of him becoming sedate will be undesirable. Looking at video of a coronavirus testing strategy press conference held on September 28, it appears he may have had a slight cold. This was not apparent in the debate the following night. He probably became significantly infectious about 16 hours before first symptoms became apparent.

Elevated IgM Causal Relationship With Severe Cases / Thrombosis?
Since IgM pentamer is a large molecule with multiple binding sites, is a large viral and IgM presence related to thrombosis or agglutination that may be causal to hypoxemia?
Some searches yet to be completed:
lack of IgG in serious case COVID
IgM elevated thrombosis covid
IgM elevated agglutination covid
IgM reduced steroids
IgM protagonist
“Due to its larger conformation, pentameric IgM can efficiently agglutinate erythrocytes, or cause red blood cell clumping, a process referred to as hemagglutination.”
“Unlike IgG or IgE, multimeric IgM does not readily enter the tissues from the bloodstream due to its larger size.”
“However, its high valency can allow it bind to antigens with a wide range of avidities (10-3 to 10-11 M-1; average values range between 10-6 and 10-7 M-1) [3,4], and to cause agglutination or clumping, a process which facilitates the removal of foreign pathogens or antigens (e.g., viral particles). Indeed, IgM is 100 to 10,000 times more effective than IgG in mediating agglutination. Agglutination is considered to be a key component of the process of IgM-mediated virus neutralization, given that a single bound IgM can activate complement and lyse an erythrocyte, while a thousand or more IgG molecules are required“
From cancer research, we have learned that in Waldenstrom macroglobulinemia that “Other WM symptoms are caused by the large amounts of abnormal IgM antibody (M protein) made by the cancer cells“. These symptoms can include headache, dyspnea (shortness of breath), and myalgia (muscle / body aches). A macroglobulin is a plasma globulin of high molecular weight which can include IgM. Studies of migraine headaches have found associations between “Elevated immunoglobulin levels and complement activation“. An article on Immunoglobulin Therapy states “Headaches associated with Ig are not uncommon and may occasionally be severe, especially in patients with a history of migraine headaches. These headaches may occur during the infusion or as long as three days later.” Also related could be cryoglobulinemia – and these symptoms can include “In people with cryoglobulins, exposure to cold temperatures can lead to pain, itching, a bluish color, or even sores on the tip of the nose, ears, fingers, or toes due to reduced blood flow to these areas.” which might be related to covid toes. It is notable that mAb therapy that was approved in an FDA EUA in November 2020 stated “Casirivimab and imdevimab are not authorized for patients who are hospitalized due to COVID-19 or require oxygen therapy due to COVID-19” which could suggest that the mAb therapy could complicate existing dyspnea – would have to look at the trial details to better understand this possible link. A specific case of a 60-Year-Old Woman With Headache and Blurred Vision stated “Particularly high-serum IgM levels can result in a hyperviscosity crisis characterized by headaches, blurred vision, epistaxis, retinal hemorrhage, intracranial hemorrhage, and altered mentation.” Another study on Adverse Effects of Immunoglobulin Therapy found various adverse effects including “flushing, headache, malaise, fever, chills, fatigue and lethargy, are transient and mild. However, some rare side effects, including renal impairment, thrombosis, arrhythmia, aseptic meningitis, hemolytic anemia, and transfusion-related acute lung injury”

IgM timeline:
https://pubmed.ncbi.nlm.nih.gov/15606632/
https://www.grepmed.com/images/7831/interpretation-antibodies-laboratory-sarscov2-timeline-serology-antibody
https://jamanetwork.com/journals/jama/fullarticle/2770758
Hypoxemia / hypoxia:
https://www.nytimes.com/2020/04/30/well/live/coronavirus-days-5-through-10.html
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7397783/
https://www.nejm.org/m/doi/story/10.1056/feature.2015.09.30.47?page=-15
https://www.sciencedirect.com/science/article/abs/pii/S0025619620303670
https://respiratory-research.biomedcentral.com/articles/10.1186/s12931-020-01462-5
https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(20)30232-2/fulltext
Many sources that agree with “Chronological” and many that have a different timeline for IgM. It seems like there are 2 “camps”. The “textbook” camp shows IgM occurring early and well before IgG but this paper (and other “lab measured”) show them nearly simultaneous and even IgG before IgM. The “textbooks” show IgM ramp beginning as early as 0 days PSO or within a week. “Antibody kinetics” shows an interesting table where test sensitivities are different for days < 14 vs days > 14. Examining why these differences exist, it may be observed that ELISA is based on binding affinity. So if “average” affinity of IgM is growing over time, it could explain why “measured” titer is later than “textbook”. It was also found that the following suggests IgM affinity grows during somatic hypermutation:
https://www.nature.com/articles/s41598-018-32926-w
More references:
“Antibody kinetics”
https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0240395
“IgG-and-IgM … timeline
“https://ppemedical.com/blog/covid-19-coronavirus-is-there-a-cure-for-the-common-cold/igg-and-igm-antibody-levels-and-symptoms-timeline/
COVID-19 STAGES, TARGET TIMELINES AND CLINICAL
https://www.confirmbiosciences.com/wp-content/uploads/2020/04/coronavirus-testing-explained-infographic.pdf
” IgM antibodies to SARS-CoV-2 are generally detectable in blood several days after initial infection, although levels over the course of infection are not well characterized”
https://www.fda.gov/media/136625/download
“Between each step, the plate is typically washed with a mild detergent solution to remove any proteins or antibodies that are non-specifically bound.”
https://en.wikipedia.org/wiki/Elisa Montalenti
The first antibodies to be produced in a humoral immune response are always IgM, because IgM can be expressed without isotype switching (see Figs 4.20 and 9.8). These early IgM antibodies are produced before B cells have undergone somatic hypermutation and therefore tend to be of low affinity.
https://www.ncbi.nlm.nih.gov/books/NBK27162/
IgM titer may be occurring earlier, but it may be so non-specific that the ELISA and other binding-based tests don’t measure the real quantity in blood? But it’s there? And hypothetically adding an agglutination issue to the mix of madness. And maybe an early one that exacerbates other issues such as hypoxemia and virally caused cellular damage.
https://www.thermofisher.com/us/en/home/life-science/antibodies/antibodies-learning-center/antibodies-resource-library/antibody-methods/immunoglobulin-igm-class.html
https://www2.palomar.edu/users/warmstrong/aniblood.htm
https://onlinelibrary.wiley.com/doi/full/10.1111/j.1751-2824.2008.00185.x
https://dm5migu4zj3pb.cloudfront.net/manuscripts/111000/111814/cache/111814.1-20140626150524-covered-253bed37ca4c1ab43d105aefdf7b5536.pdf
IgM titer may be occurring earlier, but it may be so non-specific that the ELISA and other binding-based tests don’t measure the real quantity in blood? But it’s there? And hypothetically adding an agglutination issue to the mix of madness. And maybe an early one that exacerbates other issues such as hypoxemia and virally caused cellular damage.
https://www2.palomar.edu/users/warmstrong/aniblood.htm
https://onlinelibrary.wiley.com/doi/full/10.1111/j.1751-2824.2008.00185.xhttps://dm5migu4zj3pb.cloudfront.net/manuscripts/111000/111814/cache/111814.1-20140626150524-covered-253bed37ca4c1ab43d105aefdf7b5536.pdf
Regarding sizes:
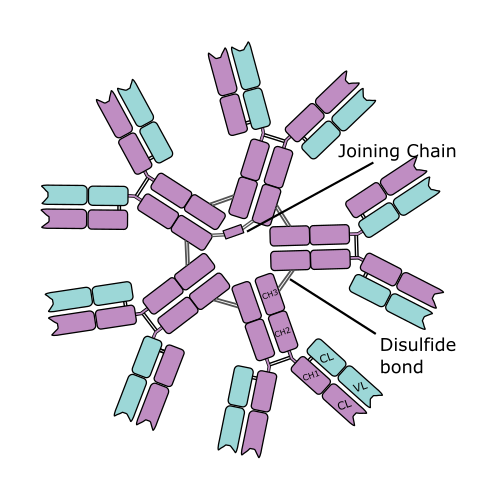
IgM pentamer diameter: about 50nm from a electron micrograph image
https://www.pnas.org/content/106/35/14960#:~:text=Cryo%2DAFM%20of%20the%20Human%20IgM%20Pentamer.&text=3.,13%20%C2%B1%203%20nm%20wide.
Capillary diameter: approximately 5 micrometers
Capillaries are very thin, approximately 5 micrometers in diameter, and are composed of only two layers of cells—an inner layer of endothelial cells and an outer layer of epithelial cells. They are so small that red blood cells need to flow through them single file.
https://www.verywellhealth.com/what-are-capillaries-2249069#:~:text=Capillaries%20are%20very%20thin%2C%20approximately,flow%20through%20them%20single%20file.
Red Blood Cell diameter: about 7x3x1 nm
A typical human red blood cell has a disk diameter of approximately 6.2–8.2 µm[18] and a thickness at the thickest point of 2–2.5 µm and a minimum thickness in the centre of 0.8–1 µm
So, not sure how something 7x3x1 um squeezes through a 5um diameter capillary, maybe it deforms the capillary to be oval as it passes? So let’s just go with the 5um diameter capillary and a 0.05 um IgM. This means that IgM is about 1% the size of the capillary diameter. If there were 100 of them clumped together they would clog a capillary. Add RBC’s and they could create some problems?
“It is present in high concentrations in blood (in the range of 1.5mg/ml)”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2747358/#:~:text=Properties%20of%20IgM%20important%20for%20microbial%20immunity&text=It%20is%20present%20in%20high,response%20following%20immunization%20or%20infection.
And right now I don’t feel like doing the math on that to convert mg to quantity per mL and then converting/scaling mL to physical size of capillary. Ok, here we go:
1 mL = 1 cc, so there are 1e12 cubic um per cc
5um diameter sphere volume is 65 cubic um. (sphere volume not needed in calculation, just sphere diameter)
IgM molecular weight is 970 kDa https://www.sciencedirect.com/topics/medicine-and-dentistry/immunoglobulin-m
1.5mg is 9.0332e+17 kDa, so that means there are about 9e+14 IgM pentamers per 1mg
So there are about 9e+2 or 900 pentamers per cubic um; call it 1000 and that’s about 10 (cube root) pentamer diameters per um, so that’s about 50 pentamer diameters per capillary diameter
And since the IgM pentamer diameter is about 50 nm, that’s about 2500 nm worth of pentamer diameters trying to fit through a 5000 nm capillary diameter. It fits, but add RBC’s and things get tight
This is looking like it supports the theory of an IgM log-jam in the capillaries. Tissues get starved. Alveoli get blocked. Most tissue survives but some dies. Hypoxemia. Covid toes. Do toes have smaller capillaries or otherwise already starved for blood flow?









Severe Immunosuppression From Aggressive Treatment?
“27 COVID-19, 51 septic, 18 critically-ill non-septic (CINS)” The 51+18 group was screened to avoid “greater than or equivalent to 300 mgs/day of hydrocortisone or other immunosuppressive medications” but the COVID-19 cohort was not screened, implying they could have been hit hard with steroids?
Separately, treatment data in other articles, COVID treatments of 6mg/day dexamethasone which appears equivalent of 150mg hydrocortisone but longer half-life? >36hrs? So it would accumulate up over days? If that were the treatment (again – no way to know from this publication?) how would that end in 10 to 15 days? Is that too much steroid? Is a short hit and pause a better treatment? Or is this study correct and it’s not cytokine induced inflammation at all? Or is it excess treatment? Is short half-life ICS Budesonide a less aggressive and improved treatment in some cases?

Either the virus or the body is purposely causing hypoxemia as part of a pathogen response.
Is the virus something novel that can improve immune response to cancer or other pathogens?
Did a virus become a retrovirus that amplifies rather than defeats CD4 T helper cells thus increasing cytokine production?
Did a virus become a retrovirus that impacts red blood cells rather than leukocytes?
Is the body optimizing immune response to kill the novel viral pathogen?
“these doctors are saying their patients behave more like they have high altitude sickness than a viral infection.”







search hypoxia inhibits cd4 t helper
