Antibody test data helps us understand how many “unreported” cases exist (people that had it and didn’t get tested; people who didn’t understand what they had was COVID19; people who felt nothing but had it). Unreported cases can make fatality rates appear higher than reality. Antibody tests can help us also understand what percentage of a population has already recovered and how far up the “herd immunity” curve the population has become. Generally, the more recovered in a population, the less likely it is that an uninfected person will become infected because those with immunity do not make good transmitters of the virus.
The antibody test data below appears to show Infection Fatality Rates (IFR) of COVID19 are in the range of 0.12% to 0.58% which can be compared with Dr. Fauci’s quote of 0.1% for seasonal flu. This is far lower than some early COVID19 mortality estimates of 3%.
The data also shows that millions in the US have already recovered from COVID19 but were never tested for viral presence when they were sick and therefore were not captured by the PCR viral presence test statistics we see everyday. The data suggests that the actual number of US recovered could be somewhere in the range of 4 to 30 million as of late April 2020.
This viewpoint assumes the antibody studies were reasonably correct and the results are not radically different from the unknown realities at other major US population-centers / cities. NOTE: This data was last reviewed early May. It is subject to change as more studies are performed and improvements in existing studies are made.
| Antibody Study Location | COVID Positive Antibody Study Result Extrapolation Error Band | COVID Positive Antibody Result Fraction (Error Bounded to Extrapolated Study Population) | Study Population Adult Recovered Extrapolation | US Wide Adult Recovered Extrapolation (not properly error bounded) | Approx. Confirmed Cases Positive PCR Viral Presence Test Around Study Date | Confirmed To Extrapolated Multiplier | Approx. Deaths As Of Study Date | Approx. Mortality Rate (given adjustments from study) | Sources / References | Test Supplier | Test Specificity (lower value means more false positives) |
| LA County 1 | Low | 0.028 | 221000 | 6132000 | 7994 | 27.6 | 600.0 | 0.00271 | http://publichealth.lacounty.gov/phcommon/public/media/mediapubhpdetail.cfm?prid=2328&fbclid=IwAR23Mk08UdYZo43oqgt3S6mi1V-KlNgu0a4BC-V8LOq4miBt5THfzC5D-IE | Premier Biotech / Hangzhou Biotest Biotech | 95-99% |
| Median | 0.041 | 331500 | 8979000 | 7994 | 41.5 | 600.0 | 0.00181 | ||||
| High | 0.056 | 442000 | 12264000 | 7994 | 55.3 | 600.0 | 0.00136 | ||||
| Stanford / Santa Clara 1 | Low | 0.018 | 48000 | 3942000 | 956 | 50.2 | 100.0 | 0.00208 | https://www.medrxiv.org/content/10.1101/2020.04.14.20062463v1.full.pdf?fbclid=IwAR2ZQe5qCvv6xCmCNykxPcNA1HpOV58yulyQYdeurU4twfc4Qx6OjLDVPDs | Premier Biotech / Hangzhou Biotest Biotech | 95-99% |
| Median | 0.0249 | 64500 | 5453100 | 956 | 67.5 | 100.0 | 0.00155 | ||||
| High | 0.0317 | 81000 | 6942300 | 956 | 84.7 | 100.0 | 0.00123 | ||||
| New York 1 | Low | 0 | 15700.0 | https://coronavirus.health.ny.gov/system/files/documents/2020/04/nysdohhcpwebinar4.23.20.pdf | Wadsworth Center’s Assay SARS-CoV-2 IgG | ||||||
| Median | 0.139 | 2700000 | 30441000 | 257000 | 10.5 | 15700.0 | 0.00581 | ||||
| High | 0 | 15700.0 | |||||||||
| Miami Dade 1 | Low | 0.044 | 123000 | 9636000 | 10600 | 11.6 | 900.0 | 0.00732 | https://www.miamidade.gov/releases/2020-04-24-sample-testing-results.asp | ? | |
| Median | 0.06 | 165000 | 13140000 | 10600 | 15.6 | 900.0 | 0.00545 | ||||
| High | 0.079 | 221000 | 17301000 | 10600 | 20.8 | 900.0 | 0.00407 | ||||
| Chelsea Mass 1 | Low | 0 | 712 | 0.0 | 39.0 | https://www.foxnews.com/science/third-blood-samples-massachusetts-study-coronavirus | BioMedomics | 91% | |||
| Median | 0.32 | 12851.2 | 70080000 | 712 | 18.0 | 39.0 | 0.00303 | ||||
| High | 0 | 712 | 0.0 | 39.0 |
Peer reviewed and formally published LA County antibody seroprevalence study

Testing Overview – 2 VERY different types of tests.
PCR viral presence (nasal / throat mucous swab) tests, when positive, indicate that the virus is present in the mucous swab. This likely means the person is infected unless they are lucky enough to just have a random virus in their mucous but not actually in their body (not likely). Sometimes people who have recovered will have mucous remnants that contain viruses which may be the cause of some “had COVID, recovered, tested negative, then later retested positive again” cases. The PCR viral presence test uses an “amplification” technique that enables the most sensitive equipment to detect fewer than 50 viruses in a sample.
Antibody (blood-sample) serology tests, when positive, indicate that a person has antibodies. Antibodies form very early during infection, usually becoming measurable right around the time symptoms occur and can remain at measurable levels for years after the infection is gone and recovery is complete such that the person has had an acquired immune system response (the part of the immune system that learns how to target specific viruses and “remembers” them). If a person has no symptoms (and/or no PCR viral presence test positive), they are likely in the “recovered” state, have measurable antibodies, and those antibodies likely afford some level of future immunity. Antibodies are what enable someone to recover and many people have had such recoveries. If people still have antibodies well after having recovered, it would be exceptionally odd to think that those antibodies will not perform some immune response as they did in during recovery from the first infection.
According to the following article regarding military COVID-19 cases, it appears that the generally healthy military population had a hospitalization / serious-case rate of about 2.8% and a mortality rate of about 0.09% (3/(6221*0.55). Again these figures are for a generally healthy military population that does not have any part of it’s population on hospice and otherwise in a category of pre-active dying / old-age. The military population may contain some with life-long asthma preconditions but relatively few of the other COVID-19 high risk groups. Data for this analysis is taken from the following article.

Also see Daily Reporting Is Misleading.
If the number of reported deaths are inflated as suspected, the mortality rates stated above need to be adjusted downward accordingly.
Antibody Test Information.
The Abbott Antibody Rapid Test Kit appears to have excellent specificity.
Only tests with very high specificity are accurate enough to ensure a false positive result does not occur. A false positive could occur from an antibody to a virus besides SARS-CoV-2 triggering a positive. This could falsely lead someone to believe that they are immune to SARS-CoV-2 / COVID19 when really they are immune to some other virus. High specificity means that the test ONLY detects antibodies to the specific virus that the test targets.
The simple rapid test kits lack sensitivity to find antibodies in people who recovered long ago from their infection (>20 to 90 days). So a negative result from these rapid test kits does NOT mean that antibodies do not exist in the blood sample. It does mean that there were insufficient quantities to detect with that inexpensive test. A lab grade, high sensitivity, “titrate”, quantitative test is needed to test for older infections. For example, if you think some flu symptoms you had 2, 3, or more months ago may have been COVID19, the rapid test kit finger-prick test will probably not detect it. You will need a higher lab grade “titrate” quantitative high-sensitivity test.
And again, to ensure that the test is highly SPECIFIC only to SARS-CoV-2 / COVID19, the test must have very high specificity.

Assure COVID-19 IgG/IgM Rapid Test Device – Assure Tech. (Hangzhou Co., Ltd)
“Positive results may be due to past or present infection with non-SARS-CoV-2 coronavirus strains, such as coronavirus HKU1, NL63, OC43, or 229E”.
Is this the same as the BD Biomedomics?










ELISA method may be more sensitive than CLIA method in detecting old infections. Certainly the finger-prick lateral flow rapid test kits are not sensitive enough to detect older infections (> 30 to 60 days).

The study below appears focused on “near date of infection” results. Need a study that is “several months after infection”.



Quest has good info on their site and may be among the best?
It appears their Abbott test is a CLIA type which may not be as sensitive as an ELISA type test (see above). They also have tests from 2 other manufacturers. Still researching.

The Solution appears to use the Abbott test which has low false positives (good specificity), but perhaps less than best sensitivity such that it cannot detect recovered infections that occurred more than 1 or 2 months prior.

While this test data is not well reviewed, the average fatality rate from all these test results is about 0.33% which correlates to the better reviewed but narrower statistical sample data shown near the top of this page.
| Study/Release | Test Type | Infected (%) | Undercount/ Ascertainment Bias | Regional IFR (%) | Population | Link |
| Northern France Study | Serological | 25.9 | 582 | 0 | Oise high school students | https://www.medrxiv.org/content/10.1101/2020.04.18.20071134v1 |
| Gibraltar Study | PCR | 2 | 7 | 0 | Gibraltar Region | https://www.youtube.com/watch?v=FnlMRUWSX2Q&fbclid=IwAR1UO7j5YD_QQnaBwCJRyk1fxr1j0_Vkr7pLJCmqvbvXHPt1FMB82BpYr0U |
| San Miguel Sample | Serological | 2 | 15 | 0 | Sample of whole Colorado town | https://www.sanmiguelcountyco.gov/CivicAlerts.aspx?AID=511&fbclid=IwAR3XfQkv1OCSuOY-w1FkDCu0BDGnG07yf-D9_Ilne_Bd8oYAnJ80S6w2QZU |
| LA County Flu Surveillance | PCR | 5 | 100 | 0.01 | LA Flu Surveillance | https://jamanetwork.com/journals/jama/fullarticle/2764137?guestAccessKey=525fc308-4cd3-4670-ad73-15b8b019f2a8&utm_source=For_The_Media&utm_medium=referral&utm_campaign=ftm_links&utm_content=tfl&utm_term=033120&fbclid=IwAR002a6aaBUV-_4OMIzIofWAjp_DAbB8hZejGVA5BLOvrEUR68tVb1prP8A |
| International Medical Cluster Study | Serological | 10 | 50 | 0.03 | Moscow Residents | https://www.reuters.com/article/us-health-coronavirus-russia-antibodies/up-to-one-in-10-residents-of-moscow-may-have-had-coronavirus-infections-laboratory-idUSKCN2262Z7?utm_source=reddit.com |
| Flu-like Surveillance | PCR | 10 | 100 | 0.05 | Flu Surveillance | https://www.medrxiv.org/content/10.1101/2020.04.01.20050542v2?fbclid=IwAR0ccDKohage1txFGvJ7kSZavLbhx1xUg_-UcxT_Kjq43rAKXh2GB-0Ttxc |
| Pregnant Sample | PCR | 7 | 143 | 0.05 | Pregnant Women in Sweden | https://www.svt.se/nyheter/inrikes/coronatestar-alla-gravida-pa-forlossningen-sju-procent-smittade?fbclid=IwAR2nGl4OLF0rduQVKr3YF4C1K54s45jIym8Y0eEpQTNlfriIkGKrDveCcG0 |
| Denmark Study | Serological | 70 | 0.05 | Capital Region | https://www.sst.dk/-/media/Udgivelser/2020/Corona/Status-og-strategi/COVID19_Status-6-uge.ashx?la=da&hash=6819E71BFEAAB5ACA55BD6161F38B75F1EB05999&fbclid=IwAR0yF9-CVqtvldch1wRB0d2esxDwzH-rjv3XRJq62Qt1WWAkR48BIMauvbI | |
| Kobe Serology Study | Serological | 2.7 | 627 | 0.06 | Kobe, Japan | https://www.medrxiv.org/content/10.1101/2020.04.26.20079822v1.full.pdf |
| Danish Blood Donor Study | Serological | 1.7 | 11 | 0.08 | Danish Blood Donors < 70 years old | https://www.medrxiv.org/content/10.1101/2020.04.24.20075291v1 |
| Homeless Sample | PCR | 36 | 37 | 0.08 | Boston Homeless | https://www.wbur.org/commonhealth/2020/04/14/coronavirus-boston-homeless-testing?fbclid=IwAR0Ai5luXYXKOGLB8Y-znlk0yXakg5Q_R_mowEV1bU8AGt15VaC2a99Ft7g |
| Swedish Study | PCR | 2.5 | 20 | 0.08 | Stockholm | https://www.folkhalsomyndigheten.se/nyheter-och-press/nyhetsarkiv/2020/april/resultat-fran-undersokning-av-forekomsten-av-covid-19-i-region-stockholm/?fbclid=IwAR2YDRAod3hXJ8CB0-2n3VB2ei30LFB3jsN6Ek9MolXitelycNv6ZpIrm-g |
| Swedish Study | Serological | 20 | 105 | 0.11 | Swedish Hospital Workers | https://www.ds.se/om-oss/press2/#/pressreleases/var-femte-medarbetare-har-haft-covid-19-2994310 |
| Iranian Study | Serological | 33 | 543 | 0.12 | Guilan province, Iran | https://www.medrxiv.org/content/10.1101/2020.04.26.20079244v1 |
| Wuhan Repatriation | PCR | 34 | 46 | 0.12 | Repatriation Flights | https://www.medrxiv.org/content/10.1101/2020.02.12.20022434v2?fbclid=IwAR0xX_0fZLr2K0DCh9X8uIlbE1GJPnRpDikxBrah3UD14vRP21yI3kHDBR8 |
| Greek Repatriation Flights | PCR | 6.3 | 59.65 | 0.15 | Repatriated Barcelonians | https://academic.oup.com/jtm/article/doi/10.1093/jtm/taaa054/5820895 |
| Private SF Study | Serological | 0.5 | 10 | 0.15 | Self-Selected SF Residents | https://twitter.com/friedberg/status/1245461209542868992?s=20&fbclid=IwAR2gmBbFnTMcp4pl2d-x8nkvQDXVRB3q-4h3Og3etHmxXZ4W5-g_NbyaABE |
| Slovenia Study | Serological | 3 | 41 | 0.16 | Slovenian Population | https://www.rtvslo.si/zdravje/novi-koronavirus/prekuzenost-z-novim-koronavirusom-pri-nas-nizka/522756 |
| Idaho Study | Serological/PCR | 1.79 | 13 | 0.17 | Boise Population | https://www.medrxiv.org/content/10.1101/2020.04.27.20082362v1 |
| Santa Clara County | Serological | 2.8 | 24.5 | 0.17 | Santa Clara County | https://www.medrxiv.org/content/10.1101/2020.04.14.20062463v2 |
| Swedish Serological Study | Serological | 10 | 25 | 0.18 | Stockholm | https://www.kth.se/aktuellt/nyheter/10-procent-av-stockholmarna-smittade-1.980727 |
| Miami-Dade Study | Serological | 6 | 15 | 0.2 | Miami-Dade Residents | https://www.miamiherald.com/news/coronavirus/article242260406.html?fbclid=IwAR2svgTSw4gmNvTrCl35bG1lYQtmqkMXOc4Sre_92zLdJx1Uyr1T6FeaR2U |
| LA County | Serological | 4.1 | 41.5 | 0.2 | LA County | http://publichealth.lacounty.gov/phcommon/public/media/mediapubhpdetail.cfm?prid=2328&fbclid=IwAR3MpnIeX6earYrGRwAGrMWpEr3K5MOk7zB29hgNHJW4krvpyeGJRGEHqT8 |
| SF Study | PCR | 1.4 | 7.04 | 0.25 | Mission District Latinx Population | https://www.ucsf.edu/news/2020/05/417356/initial-results-mission-district-covid-19-testing-announced |
| Iceland Continous Sampling | PCR | 1 | 0.28 | Iceland | https://www.nejm.org/doi/full/10.1056/NEJMoa2006100 | |
| Median Averages | 23.3 | 0.28 | ||||
| Super-spreader Study | PCR | 15.5 | 5 | 0.36 | Super-spreader event in Germany | https://www.ukbonn.de/C12582D3002FD21D/vwLookupDownloads/Streeck_et_al_Infection_fatality_rate_of_SARS_CoV_2_infection2.pdf/%24FILE/Streeck_et_al_Infection_fatality_rate_of_SARS_CoV_2_infection2.pdf |
| Wuhan Hospital | Serological | 10 | 22.1 | 0.36 | Wuhan Region | https://onlinelibrary.wiley.com/doi/10.1002/jmv.25904 |
| German Survey | Serological | 14 | 20 | 0.37 | Gangelt | https://www.uni-bonn.de/news/111-2020?set_language=en |
| Finnish Institute | Serological | 3.4 | 6 | 0.39 | Helsinki | https://thl.fi/en/web/thlfi-en/-/number-of-people-with-coronavirus-infections-may-be-dozens-of-times-higher-than-the-number-of-confirmed-cases?fbclid=IwAR3bDYNRIQBQFxhJdrmjypoHtbk_thqb5E_ayR9IUU3FUGeXrP3oc6GwpS0 |
| Italian Epidemic Report | PCR | 10.7 | 30 | 0.4 | Italian Healthcare Workers | https://www.epicentro.iss.it/coronavirus/bollettino/Bollettino-sorveglianza-integrata-COVID-19_16-aprile-2020.pdf#page=13 |
| Switzerland Study | Serological | 5.5 | 10 | 0.4 | Geneva | https://www.hug-ge.ch/medias/communique-presse/seroprevalence-covid-19-premiere-estimation |
| Second Switzerland Study | Serological | 9.7 | 9.55 | 0.5 | Geneva | https://www.medrxiv.org/content/10.1101/2020.05.02.20088898v1.full.pdf |
| Total COVID-19 Mortality in Italy | PCR | 67 | 35 | 0.5 | Italy extrapolated to NYC | https://www.medrxiv.org/content/10.1101/2020.04.15.20067074v2 |
| Northern France Study | Serological | 3 | 69 | 0.5 | Oise blood donors | https://www.medrxiv.org/content/10.1101/2020.04.18.20071134v1 |
| Pregnant Sample | PCR | 14 | 17 | 0.53 | Pregnant Women in NYC | https://www.nejm.org/doi/full/10.1056/NEJMc2009316?fbclid=IwAR0MOuMrCb3xe0lfiNMZwND4FN_HSDimM_fLdBrmDXuWeJzLVc4bnKlH48M |
| London HCW Screening | PCR | 7.1 | 35 | 0.54 | London Health Care Workers | https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)31100-4/fulltext |
| Madrid Study | PCR | 11.6 | 31 | 0.54 | Madrid Hospital Workers | https://www.medrxiv.org/content/10.1101/2020.04.07.20055723v1?fbclid=IwAR0mg2DXxFZUNuzYuGuQOkwgQySH5_bUpwODJdWeWFONjCAbheml28hxPCs |
| HCW Madrid Study | Serological/PCR | 11.2 | 31 | 0.56 | Madrid Hospital Workers | https://www.medrxiv.org/content/10.1101/2020.04.27.20082289v1 |
| New York Study | Serological | 13.9 | 11.2 | 0.56 | New York State Residents | https://www.governor.ny.gov/news/amid-ongoing-covid-19-pandemic-governor-cuomo-announces-statewide-antibody-testing-survey-will |
| Ohio Prison Data | PCR | 66 | 0.57 | Marion Correctional Institution | https://drc.ohio.gov/Portals/0/DRC%20COVID-19%20Information%2005-03-2020%201253.pdf | |
| Czech Study | Serological | 0.4 | 5 | 0.59 | Czech Population | https://medicalxpress.com/news/2020-05-czech-covid-incidence-population.html |
| Covid-19 Simulation | PCR | 0.6 | Repatriation Data | https://www.medrxiv.org/content/10.1101/2020.03.09.20033357v1.full.pdf?fbclid=IwAR3ATHWmq2CQRMGy95V-YV6LZ4hBZcXt2WJuKhmEBxuvcoHY9mdNM-HsB8E | ||
| Dutch Survey | Serological | 3 | 31 | 0.67 | Netherlands | https://www.researchsquare.com/article/rs-25862/v1 |
| Chelesa Data | Serological | 33 | 30.1 | 0.71 | Chelesa Street Survey | https://www.bostonglobe.com/2020/04/17/business/nearly-third-200-blood-samples-taken-chelsea-show-exposure-coronavirus/?fbclid=IwAR3XQlYzGz7cMfc3nmQSAbG8jTICJ7nkG8ZMfH-fObf8QmQ2FYthvSiAEkQ |
| Continous New York State Study | Serological | 19.99 | 10.06 | 0.77 | New York City Residents | https://www.governor.ny.gov/news/amid-ongoing-covid-19-pandemic-governor-cuomo-announces-results-completed-antibody-testing |
| Early French Analysis | PCR | 8 | 0.8 | Simulation | https://www.medrxiv.org/content/10.1101/2020.03.22.20040915v4 | |
| Italy Town Sample | PCR | 70 | 10 | 1 | Sample of whole Italian town | https://www.lastampa.it/topnews/primo-piano/2020/04/02/news/coronavirus-castiglione-d-adda-e-un-caso-di-studio-il-70-dei-donatori-di-sangue-e-positivo-1.38666481?fbclid=IwAR2w8-93Obhq4uRHpZYbbn-yEO_LjmCPMtQdXQ-wkkmCInIj9rxXJeEAwJc |
| Italian Analysis | PCR | 5 | 1 | Repatriation Data | https://www.thelancet.com/journals/laninf/article/PIIS1473-3099(20)30227-9/fulltext?fbclid=IwAR3b9eOpxeQLbUxmxjFHGK2b2M2tM7dRerS0Y5cWPV6H6B1QPqZsN_GzDX0 | |
| Ohio Prison Data | PCR | 73 | 1.44 | Pickaway Correctional Institution | https://drc.ohio.gov/Portals/0/DRC%20COVID-19%20Information%2005-03-2020%201253.pdf | |
| Scottish Blood Donors | Serological | 0.6 | 20 | Scotland | https://figshare.com/articles/Serological_analysis_of_1000_Scottish_blood_donor_samples_for_anti-SARSCoV2_antibodies_collected_in_March_2020/12116778/2?fbclid=IwAR1wkF1QVZeAt6jDNmeWVJwBmK6Bo8zYPnBRJruvfOYqYO9qFEXuMndjpI8 | |
| Wuhan Flu Surveillance | PCR | 1.4 | 20 | Wuhan Flu Data | https://www.nature.com/articles/s41564-020-0713-1 | |
| Covid-19 Simulation | PCR | 5 | Simulation | https://science.sciencemag.org/content/early/2020/03/24/science.abb3221.full?fbclid=IwAR3NQ_ybQnjzyysGQPvLsb-9zfKOoHYY22CJMMbry7-mCGEC1LYcTjQf0Z0 | ||
| 17.69 | 0.333774 |
Ascertainment Bias,Regional
IFR (%),Population,Link
London HCW Screening,PCR,7.10,35.00,0.54,London Health Care Workers,<a href=

In a small population of generally healthy navy sailors, as of mid-May, 1 death occurred in 1156 PCR viral presence test confirmed positive cases, resulting in a mortality rate of 0.087%. If another death occurs, that would double to 0.17%. Again, keeping in mind Dr. Fauci’s stated rate of 0.1% for seasonal flu across all population including those at higher risk. While asthma is considered a risk factor, it is not known if the navy screens out asthma sufferers and personnel with potential risk factors. But we do know that good general health is a screen for active duty.

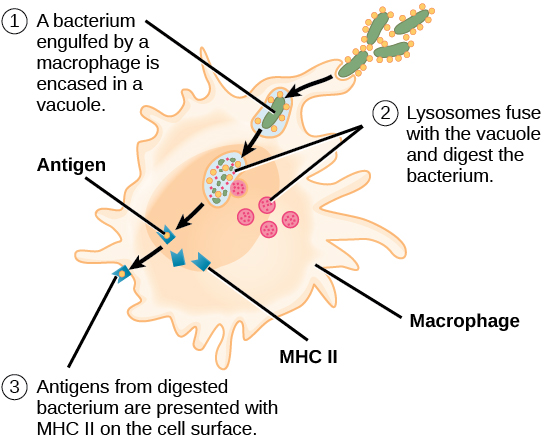
Immune System Function
Some great introductory material on the immune system. In about an hour, many people could develop a good fundamental understanding of the immune system.

After reviewing all of the Khan videos, the following videos will strengthen the fundamentals and introduce additional levels of knowledge.






Back To Some Simple Statements From WHO
The WHO’s EXTREMELY MISLEADING statement:
“There is currently no evidence that people who have recovered from COVID-19 and have antibodies are protected from a second infection” – 24 April 2020
When antibodies and specific B / T cells are present, it would be the rare case that some level of immunity is not exhibited.
For the WHO to say there is NO EVIDENCE ignores all data we have with viruses in general, the general function of antibodies and the immune system, various related coronaviruses, SARS-CoV-1, and MERS related viruses.
Antibodies are what enable someone to recover and many people have had such recoveries. If people still have antibodies well after having recovered, it would be exceptionally odd to think that those antibodies will not perform some immune response as they did in during recovery from the first infection.
The WHO statement which has been quoted vociferously is horribly misleading and creates concern about WHO’s ability to communicate accurately.
Additionally, if there was no immunity, then we would expect the millions of recovered in the US alone to be complaining of re-infections, rather than a small number of re-infections that have been documented. These small number of re-infections that have occurred appear to be statistical outliers – rare occurrences.
Also See: Immunology

Jung Eun-Kyeong, Director of the Korean Centers for Disease Control & Prevention
Findings from investigation and analysis of re-positive cases

While not able to find the underlying study at this time, this is an interesting avenue to research further. If true, it could help explain why some Asian countries which were more exposed to SARS-CoV-1 appear to possibly be more resistant to SARS-CoV-2. This is being watched.
Was this the underlying study or is there another?


Is SARS-CoV-2 similar enough to SARS-1 that immune response is of similar behavior? Probably true, so potentially useful to apply SARS-1 learning.